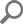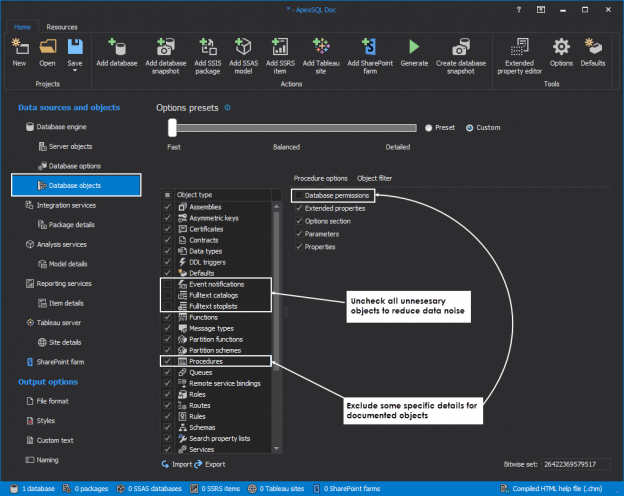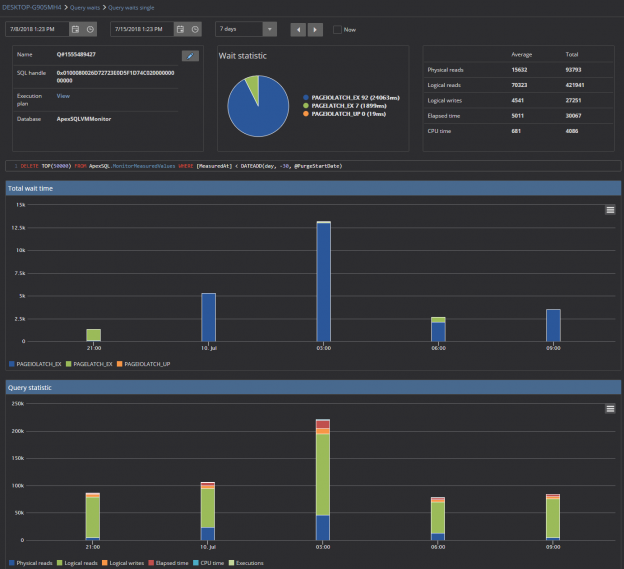
The Query waits feature is the core ApexSQL Monitor feature and, in most cases, the first step in resolving SQL Server performance issues. Query wait statistics, somewhere referred to as the Response Time Analysis (RTA), is for many less experienced or new DBAs the most complicated part of performance troubleshooting. ApexSQL Monitor’s query wait feature is designed to provide fast and easy insight in the query performance data and thus providing a significant help in identifying the causes of the SQL Server performance issues.